FeNO and peak-flow – what’s the difference?

Anyone who suffers from asthma will do everything to make the disease more manageable and thus make life easier. Measurements that can be performed at home have an important role to play here.
Conventional monitoring method: peak-flow measurement
With the help of a peak-flow meter (mechanical or digital), anyone can check their lung function at home. The device measures the maximum air flow from the lungs, or Peak Expiratory Flow (PEF). However, rather than measuring the degree of inflammation in the bronchial tubes, it shows the narrowing of the airways caused by asthma.
The measured value is recorded several times a day in a peak-flow protocol. Peak-flow measurement is not a calibrated measuring method. The values serve as a guideline only. They compare the current value with your personal best value. Knowing the current peak-flow values has been an important component of asthma management up to now. They give you an overview of the progression of the disease. And they serve your doctor as a basis for prescribing the correct medication.

FeNO measurement at home
the asthma early-warning system identifies an incipient deterioration much earlier
FeNO measurement detects how severe the inflammation in the lower airways currently is. Nitric oxide (NO), which is measured in exhaled breath during this test, is produced when the bronchial tubes are inflamed. FeNO stands for fractional exhaled nitric oxide (NO). The FeNO value is a biomarker that indicates the exact severity of the inflammation.
Two studies show benefits of FeNO measurement in therapy monitoring
A study* conducted by the University of Otage Medical School, Dunedin, New Zealand compared FeNO measurement with other asthma testing methods. The study shows that compared with lung function measurements (peak flow), FeNO measurement is much more precise. A change induced by therapeutic treatment is also indicated more reliably. It is also quick and easy to perform.
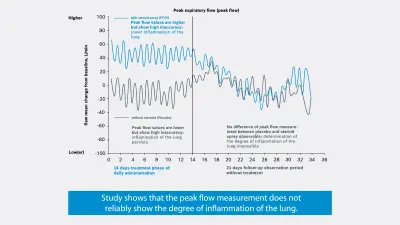
A second study** by the British pharma company GlaxoSmithKline on the effectiveness of a new steroid spray shows that as a marker, FeNO can reliably detect the degree of inflammation of the lung on administration and discontinuation of the steroid, whereas peak-flow measurement has a high inaccuracy.
What FeNO measurement can do
- It is a variable for assessing your current stamina level
- It shows the current degree of inflammation
- It shows whether the degree of inflammation is responding to medication
- It shows earlier than peak-flow measurement whether there is a risk of deterioration
- It shows earlier than peak-flow measurement whether there is a positive response to medication
The benefits of FeNO measurement for you
- The FeNO value gives you an exact and objective variable (ppb) for the degree of inflammation in your lungs
- The FeNO value helps you decide whether you need to visit your doctor
- Unlike the peak-flow meter, measuring your FeNO value does not depend on how you blow into the device. The Vivatmo me FeNO measuring device is electronically assisted to ensure the measurement conditions are always the same
- FeNo measurement does not need to be done several times a day, only several times a week
The benefits of FeNO measurement for children
FeNO measurement is above all useful for children.*** The accuracy of peak-flow measurement depends on the intensity with which you blow into the device and the exhalation speed. It is difficult for children to always perform the measurement in the same way. FeNO measurement with Vivatmo me is a standardized procedure. The results are only displayed if the breathing maneuver is successful.

Interview with Prof. Felix Herth on asthma and FeNO home measurement

Loading the video requires your consent. If you agree by clicking on the Play icon, the video will load and data will be transmitted to Google as well as information will be accessed and stored by Google on your device. Google may be able to link these data or information with existing data.
* Smith A et al. Diagnosing asthma: comparisons between exhaled nitric oxide measurements and conventional tests. Am J Respir Crit Care Med. 2004;169(4):473-8
** Peter Daley-Yates et. al., European Respiratory Journal 2017 50: OA276; DOI: 10.1183/1393003.congress-2017.OA276
*** Use of Vivatmo me is recommended for children from the age of 7
1 modified graphic - Peter Daley-Yates et. al., European Respiratory Journal 2017 50: OA276
2 FF/VI = fluticasone furoate / vilanterol